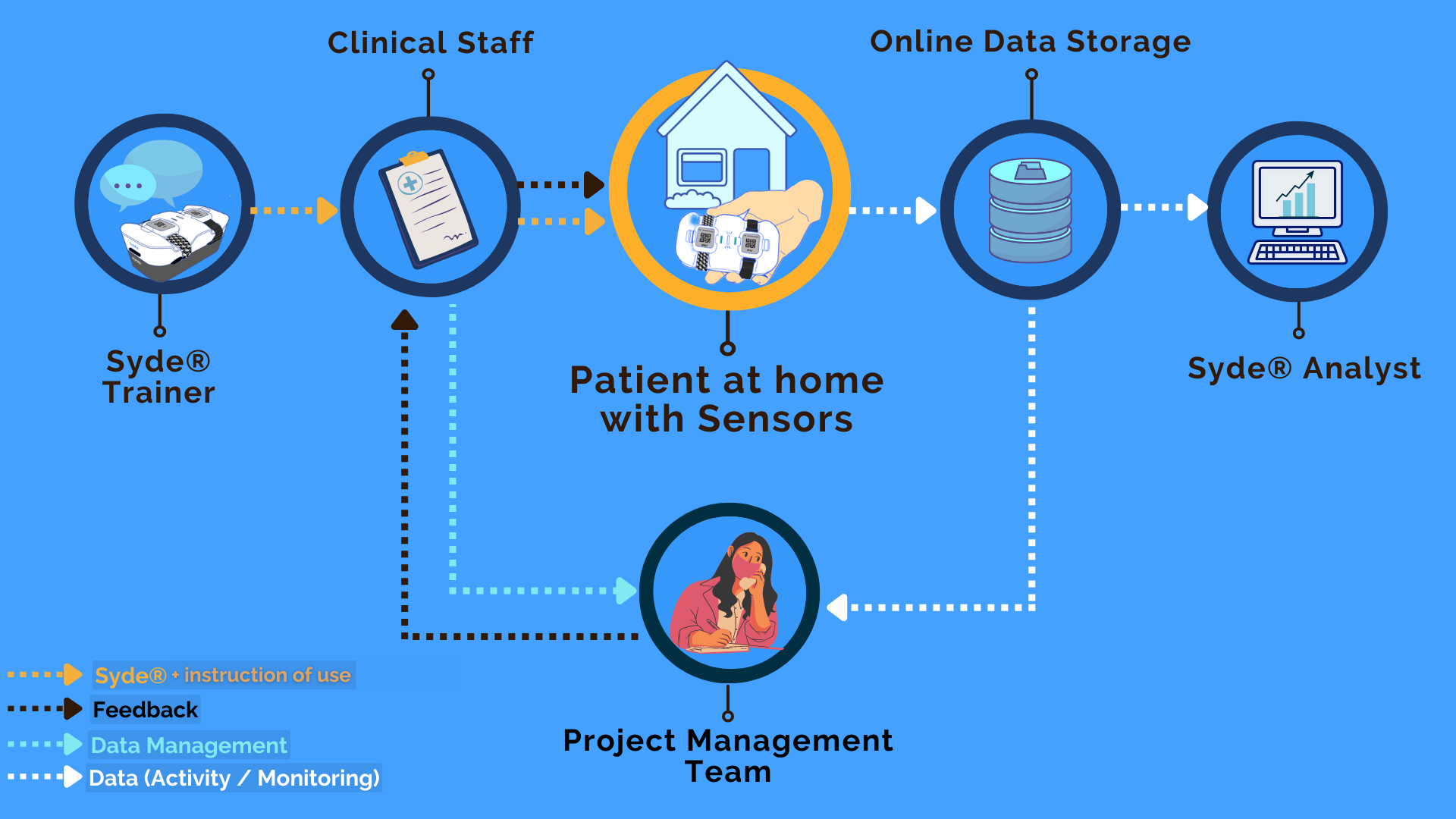
Data from wearable devices approved for use in Duchenne muscular dystrophy clinical trials | Duchenne UK
Prospective, Longitudinal study of the Natural History and functional status of patients with MyoTubular Myopathy NatHis-MTM

Data from wearable devices approved for use in Duchenne muscular dystrophy clinical trials | Duchenne UK